Advanced Software Engineering, Embedded Systems, and MedTech Technology Development for Government and Research.
Engineering solutions for custom software platforms, biomedical devices, embedded systems, and regulated technology environments.
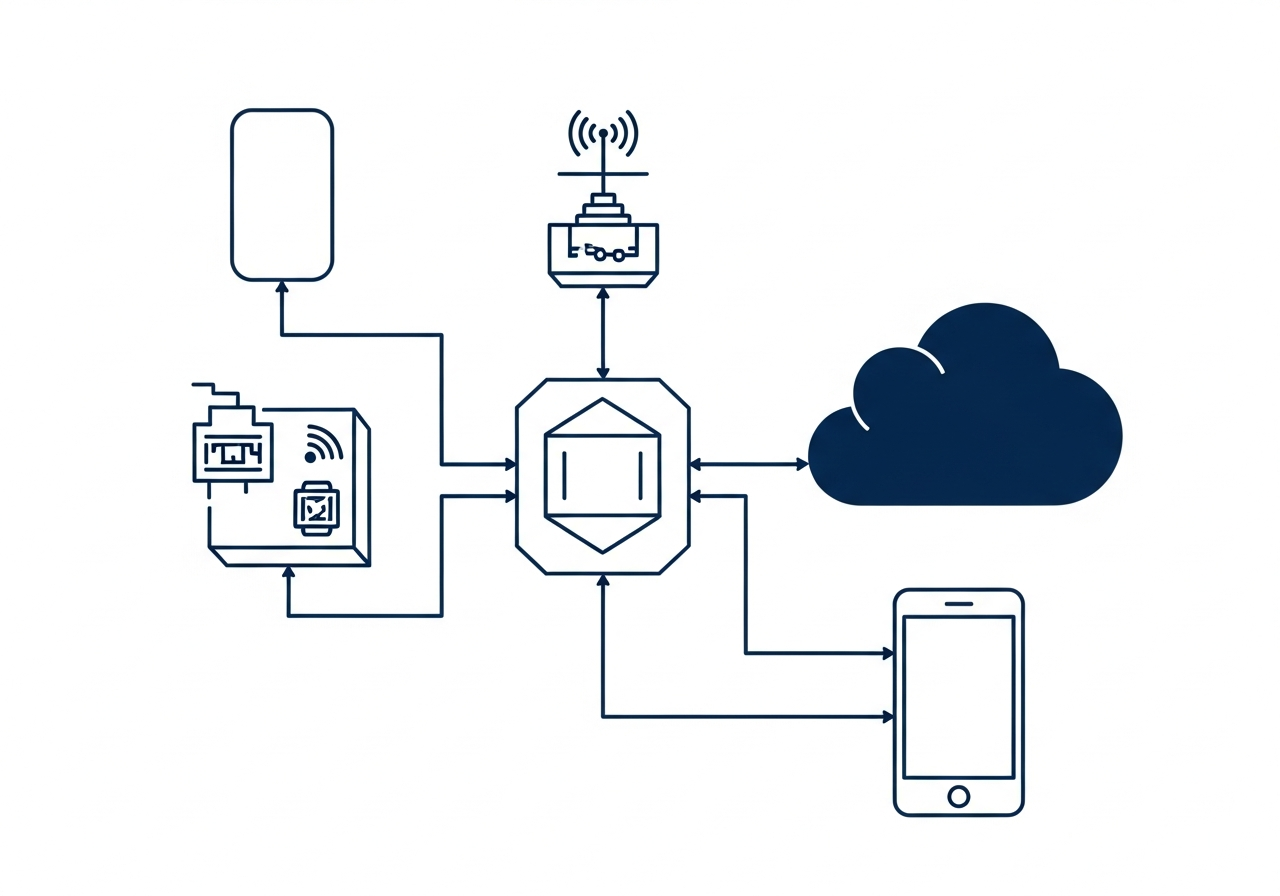
Chacheetah Inc. brings together engineering expertise developed through multiple technology and manufacturing organizations with experience in software development, embedded systems, biomedical technology, and industrial production.
Core Capabilities
- • Custom software platform development
- • Embedded systems and hardware engineering
- • Biomedical and scientific technology development
- • Research prototype engineering
- • Advanced manufacturing transition support